Raeed Deen, Andrew Bullen
|
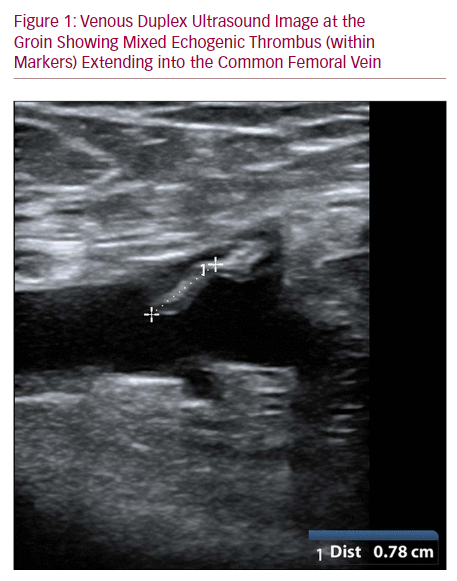
Abstract Endovenous glue ablation for lower limb varicose veins is growing in popularity due to its safety and efficacy. Of significant concern is glue-associated thrombus extension into deep veins. We present a case of thrombus extending into the common femoral vein following endovenous glue ablation for varicose veins with the VenaSeal™ closure system (VCS; Medtronic). A 63-year-old man who presented with symptomatic varicose veins had incompetence of the saphenofemoral junction. He underwent endovenous glue ablation using VCS closure. At 1 month, improvement in varicosities was mirrored by duplex ultrasound confirmation of successful long saphenous vein ablation, but ultrasound indicated thrombus extending into the common femoral vein. This was managed by surveillance duplex and serial clinical observation, with spontaneous resolution at 12 months. With increasing use of VCS for varicose veins, it is likely that thrombotic complications of the deep veins will be encountered more frequently. It is time for formulation of guideline-based management of this complication.
Disclosure: AB is on the Vascular & Endovascular Review editorial board; this did not influence peer review. RD has no conflicts of interest to declare.
Received: Accepted: Published online:
Correspondence Details: Raeed Deen, Circulation Health, Suite 101/62 Harbour Street, Wollongong, NSW 2500, Australia. E: raeeddeen30@gmail.com
Open Access: This work is open access under the CC-BY-NC 4.0 License which allows users to copy, redistribute and make derivative works for non-commercial purposes, provided the original work is cited correctly.
|
- Rasmussen LH, Lawaetz M, Bjoern L, et al. Randomized clinical trial comparing endovenous laser ablation, radiofrequency ablation, foam sclerotherapy and surgical stripping for great saphenous varicose veins. Br J Surg 2011;98:1079–87. Crossref| PubMed
- Bootun R, Lane TR, Davies AH. The advent of non-thermal, non-tumescent techniques for treatment of varicose veins. Phlebology 2016;31:5–14. Crossref| PubMed
- Lam YL, De Maeseneer M, Lawson J, et al. Expert review on the VenaSeal® system for endovenous cyano-acrylate adhesive ablation of incompetent saphenous trunks in patients with varicose veins. Expert Rev Med Devices 2017;14:755–62. Crossref| PubMed
- Kane K, Fisher T, Bennett M, et al. The incidence and outcome of endothermal heat-induced thrombosis after endovenous laser ablation. Ann Vasc Surg 2014 ;28:1744–50. Crossref| PubMed
- Dexter D, Kabnick L, Berland T, et al. Complications of endovenous lasers. Phlebology 2012;27(Suppl 1):40–5. Crossref| PubMed
- Almeida JI, Javier JJ, Mackay E, et al. First human use of cyanoacrylate adhesive for treatment of saphenous vein incompetence. J Vasc Surg Venous Lymphat Disord 2013;1:174–80. Crossref| PubMed
- Proebstle TM, Alm J, Dimitri S, et al. The European multicenter cohort study on cyanoacrylate embolization of refluxing great saphenous veins. J Vasc Surg Venous Lymphat Disord 2015;3:2–7. Crossref| PubMed
- Morrison N, Gibson K, McEnroe S, et al. Randomized trial comparing cyanoacrylate embolization and radiofrequency ablation for incompetent great saphenous veins (VeClose). J Vasc Surg 2015;61:985–94. Crossref| PubMed
- VenaSeal™ closure system. www.accessdata.fda.gov/cdrh_docs/pdf14/P140018c.pdf (accessed 20 February 2020).
